Battery Shredding - Which Method is Future Proof?
Shredding and sorting of end of life lithium ion batteries to produce black mass is an essential step in their recycling. Not only does it decommission and treat what is a dangerous waste, it also frees up valuable resources (that were trapped in old, inefficient batteries) to be reintroduced back into the supply chain. A variety of approaches have been developed, each offering their own advantages and disadvantages. A list of the most common approaches is given below.
- Shredding in nitrogen followed by vacuum drying and dry sorting
- Shredding in vacuum followed by vacuum drying and dry sorting
- Shredding under a flow of water and wet separation
- Shredding in air (under extraction) followed by dry sorting
- Thermal treatment followed by shredding (or vice versa) and dry sorting
The above systems have been engineered to avoid fires or explosions from occurring whilst the batteries are being shredded or processed afterwards. The batteries contain flammable solvents and charge which is a volatile mixture. The systems work in different ways, inert atmospheres such as those that use nitrogen or vacuum remove the oxygen from the shredding chamber. Wet processes quickly cool the material and douse it in water. Thermal treatment burns off the flammable electrolyte in a controlled environment and shredding in air under extraction removes any flammable vapour before it can reach its lower flammability limit (although this is not guaranteed to stop a fire since there is flammable solvent within the shredded material and if the extraction is not present all the way through the process, it will catch fire).
When deciding which method to choose, the recycler must take into consideration several different factors. A brief overview of the factors affecting the decision is given below:
- Safety – does the process shred the batteries safely, allowing the material inside to be accessed without causing harm to people and the environment?
- Cost of purchase – is the shredding and sorting system within the budget of the company?
- Cost of processing
- Throughput – can the plant process enough material at a sufficient rate to make money?
- Compliance – is the process compliant with the current and upcoming Regulations specifically regarding the EU Battery Directive?
- Waste generation – how much waste does the process generate and can the waste be disposed of safely and economically?
- Quality of product – does the process produce a high quality product that maximises the amount of money that can be extracted from the batteries?
- Efficiency – what are the recovery rates for the most valuable components?
- Other – such as the ability to shred full packs
Safety
The most important criteria for any process is that it should operate safely and not cause harm to any person whether inside or outside the plant. The process should have sufficient engineering controls and systems in place to ensure all hazards are contained and do not leave the plant. Hazards include fire and explosions, exposure to toxic and poisonous chemicals such as HF and metal oxides found in black mass as well as other mechanical hazards normally found on a processing plant.
Whilst fires and explosions may be the most noticeable hazards associated with lithium ion batteries, they are not the main hazards on a battery recycling plant. Recycling plants have a number of engineering and administrative controls in place to reduce the chances of fire or explosions and they are generally rare occurrences where the effects are felt immediately. The main hazards on battery recycling plants are exposure to toxic and poisonous chemicals. Some of the chemicals found in lithium ion batteries are incredibly toxic and have extremely low exposure limits. Continuous exposure to even tiny amounts can cause serious long term health problems such as cancers and organ failure. The Table below shows the exposure limits for the main components of a lithium ion battery.
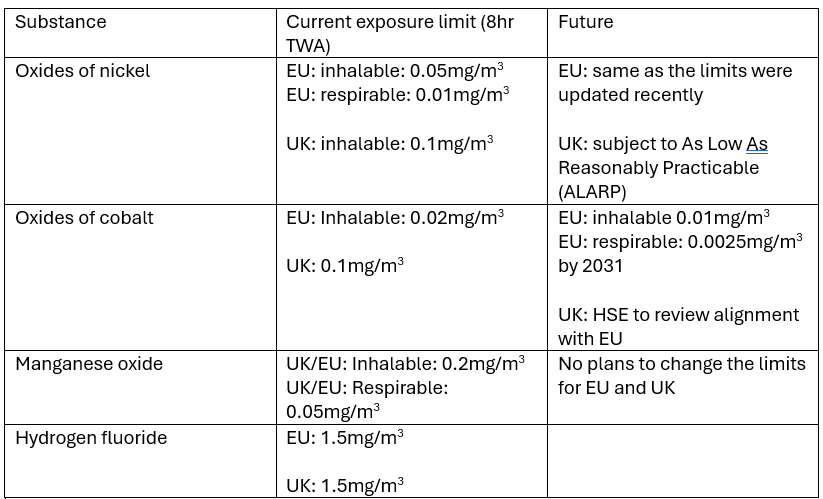
In addition to the above table, exposure to organic solvents can cause irregular injuries such as organ damage or prolonged breathing difficulties.
The EU makes a distinction between ‘inhalable’ and ‘respirable’ dust. Inhalable dust is one that can be breathed in but is large enough to be trapped in the nose hair or mucus lining. Inhalable dust can be up to 100µm whilst respirable dust is less than 5µm and passes through the body’s capturing mechanism. Once it ends up in the alveoli, it can either enter the bloodstream or can stay there forever as the body has no mechanism of removing it. Black mass contains particles that fall into both inhalable and respirable fractions. Studies have shown that up to 10% of black mass particles are less than 6.8 µm and as low as 2.7µm[1]. Sharp particles may damage the alveoli and cause similar effects to asbestos.
To illustrate to how tiny the allowable limits are, imagine a battery recycling plant inside a 10,000m3 warehouse that is 50m long, 20m wide and 10m high. Assuming the plant processes LCO chemistry, to comply with the current EU limits, the total mass of black mass allowed in the air in the whole warehouse is 0.2g for the inhalable fraction (assuming 50% LCO content of black mass). In just 5 years, this figure will dop to 0.2g for the inhalable fraction and 0.125g for the respirable fraction (assuming 10% of LCO fraction of black mass is respirable).
Anyone who has ever stepped foot on a shredding plant knows how difficult it is to be below these limits. Essentially, the only way they can be achieved is if the plant is isolated and fully automated with no human intervention. Failing that, employees need robust PPE that cover their entire bodies. Positive airflow masks equipped with the right filter types that can take out the inhalable and respirable dust as well as the HF and solvents must be mandated. Most plants use dust masks, eye protection and some form of clothing but this is insufficient as the LiPF6 present in black mass readily hydrolyses into HF in the presence of moisture (LiPF6 is hygroscopic). If black mass falls onto an uncovered body part of an employee, it will quickly turn into HF as it reacts with the moisture on the skin. If this is occurring on a daily basis, then over time, it can lead to significant exposure.
In terms of fire and explosion safety, the process must take away one or more of the following; fuel, oxygen, heat or sparks. An explosion is the ignition of a flammable gas or vapour in a confined space that generates a pressure wave. During shredding there will always be a risk of ignition, even if the battery has been fully discharged as shredding metal parts generates sufficient heat to ignite volatile vapours such as those generated from the electrolyte solvents in lithium ion batteries. Post shredding, the material quickly heats up and reaches peak temperature around 30s after the shredding. Temperature is heavily dependent on the charge in the cells and can reach over 200C post shredding.
Taking the above into account, which type of shredding and sorting process give high level of safety, meets current Regulations and is future proof for upcoming Regulations? The table below rates the different technologies based on their ability to suppress fire and explosions, contain/suppress dust generation and contain/suppress HF and solvent vapours.
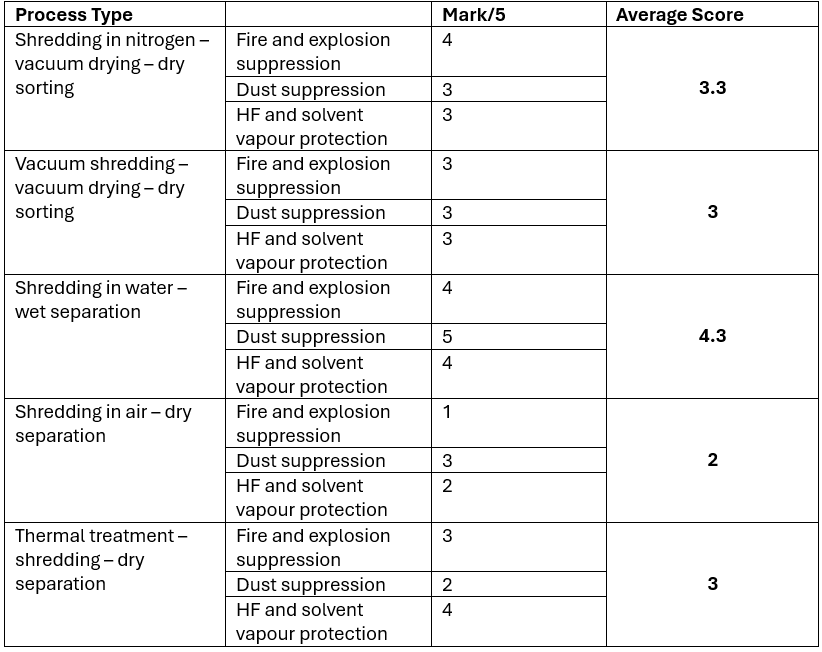
The safest method of shredding and sorting batteries is using a wet process as it provides good protection against fires and explosions (which can be further enhanced with the use of nitrogen), it suppresses dust generation as wet material cannot become airborne and it also suppresses HF and solvent vapour formation by capturing the chemicals in water. In terms of safety, it is future proof as the dust issue is likely to be the major safety concern in the future. Other processing types such as those that use nitrogen and vacuum do not suppress the dust but merely contain it and this will never be 100% efficient. For example, a plant producing 1000kg/h of black mass can have a dust containment efficiency of 99.9% but that still means 1kg of dust is released per hour (assuming that 100% of the black mass is liable to forming dust). Processes that use thermal treatment produce very dry black mass that can become airborne far more likely than black mass with some solvent inside and therefore gets a lower score in the dust suppression category.
In terms of fire and explosion suppression, processes that use vacuum operations have an inherent weakness in that air flows into the system in cases of structural failure, such as a hole forming in the shredding chamber or the vacuum dryer. The main advantages of a vacuum are that it allows the shredding to be caried out in an oxygen deficient atmosphere and reduces the boiling point of solvents, enabling their efficient recovery.
Pyrolysis or thermal treatment prior to shredding can be a good way to solve the flammability and explosion issue as it burns off the electrolyte solvents and degrades the LiPF6 but there is one issue that is often overlooked. The aluminium foil present in black mass is directly attached to metal oxides, often metals that are less reactive than aluminium. There is a chance that some form of thermite reactions can occur where the more reactive metal, aluminium, takes the oxygen from the less reactive nickel and cobalt oxides, generating extremely high temperatures that can melt through the steel reactor.
Cost of Purchase and Cost of Processing
These categories will not be marked in this article as the cost of purchase is a decision that the recycling company must make. It depends on what type of equipment they buy, who they buy it from and where they buy it. The cost of processing directly related to the throughput. Larger plants have lower costs of processing.
Other Factors
The table below marks each system based on the following factors: throughput, product quality, its ability to meet the efficiency targets as set out in the Battery Directive and waste generation.
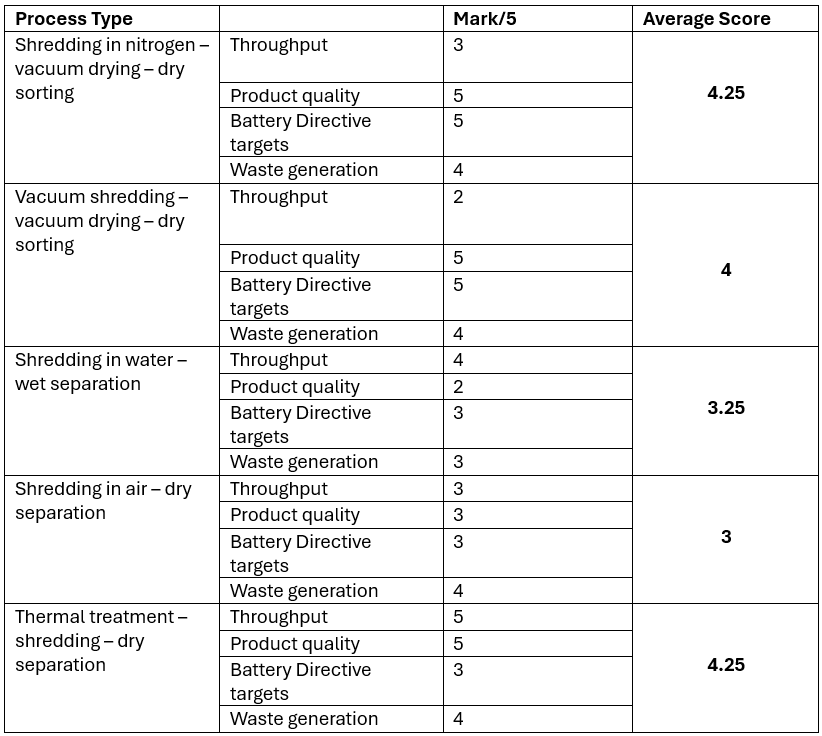
Throughput
Throughput is a function of plant size and the ability to process continuously. The ability to process continuously relies on feeding the system continuously. Systems that require an inert atmosphere or processes that operate in batch mode, such as vacuum shredding and drying, will have lower throughputs than systems that do not have such bottlenecks. Therefore, wet shredding systems can operate at a higher throughput than those that require inert atmospheres. Higher throughputs are associated with lower processing costs per tonne of material and can be scaled to tens of thousands of tonnes per year – the scale that is required in the near future. Other systems that can achieve such throughputs involve thermal treatment before or post shredding. Shredding in air can achieve continuous throughput but requires batteries to be fully discharged beforehand but they can still catch fire post shredding so require secondary systems such as fire suppression systems to allow continuous shredding.
Product Quality
Product quality is very important as it determines the price at which the black mass and other products can be sold at. The main products from shred and sort plants are copper and black mass which account for >95% of the total value in the batteries. Wet processes generate lower grade products than dry processes. Screening is a vital step in producing a high quality black mass product and it is far more difficult to screen wet black mass than it is to screen dry black mass. Wet black mass normally contains high concentrations of copper and aluminium compared to dry black mass. Some companies use filter presses to separate the water from the black mass and in doing so, introduce the solvents back into the black mass. The copper, after becoming wet, is no longer considered as ‘bright copper’ and commands a lower value than dry separated copper. All of these factors can lower the quality of the products and reduce their value. Some companies dry the shredded material post (wet) shedding and this can give a better quality product although it will lack a little bit of lithium as some of it dissolves into the water and the copper will not be bright copper.
Product quality from dry processes is generally much better as all of the lithium remains in the black mass. The solvents can be separated out for recovery and potential re-use. Thermal treatment can produce a good quality black mass but it can contain the charred remains of plastics that can lower the content of the metals per unit mass. Shredding in air with no solvent extraction step produces a black mass that has a high organic content and is potentially flammable and harder to screen as the organic content behaves like moisture and can creates clumps of black mass.
EU Battery Directive Targets
The main targets from the EU Battery Directive that affect shred and sort companies are the efficiency targets, which are currently 65%, increasing to 70% in Dec 2030. Interestingly, the shred and sort companies are also responsible for ensuring the lithium, cobalt and nickel are recovered at the rates set out in the EU Battery Directive (50% for Li, 90% for Ni, Co, Cu ad then 80% for Li and 95% Co, Cu and Ni) even though they are generally not involved in the hydrometallurgical recovery of the material. For a material to be considered recycled, it must become a product to be used in another application. Recycling does not mean claiming an 80% recycling efficiency if 1kg goes in at the front end and 0.8kg comes out at the back end. The efficiency of the downstream customers must also be accounted for until the waste becomes a product that is reintroduced into the supply chain.
Essentially it means the shredding companies must sell the copper and black mass to companies that can recover those elements at the required efficiencies. This can take the bargaining power away from the shred and sort companies and give it to the hydrometallurgical companies. The Table below shows a typical composition for an 18650 NMC 622 cell and the typical recycling efficiencies using the current system.
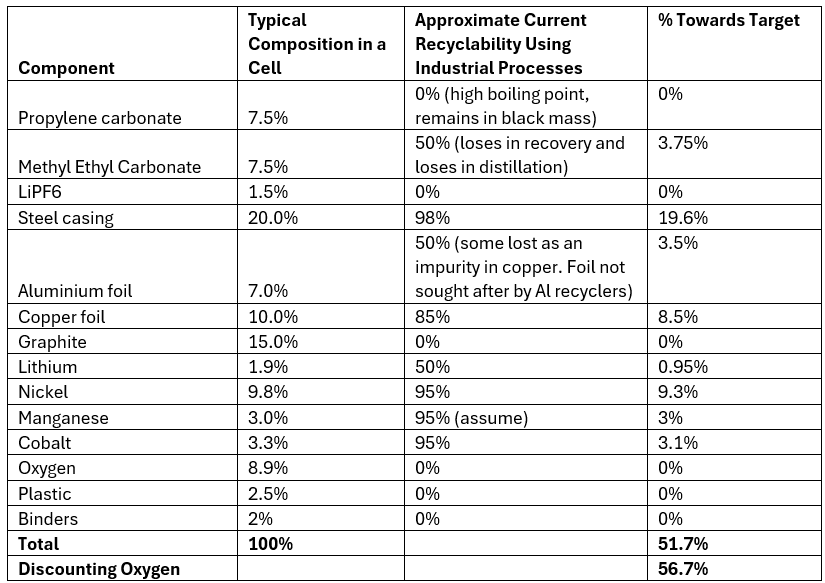
In terms of being able to meet the EU recycling efficiency targets, process that shred the batteries and then recover the electrolyte are best placed to meet the targets as theoretically they can recover all the components within the batteries, however, practically and economically, this is not feasible. Achieving the current 65% target is not easy, especially for portable batteries where there is no large pack casing that can make up a significant portion of the target percentage.
In the example above, a 56.7% recycling efficiency is achieved by the process. This is still far short of the 65% requirement. For EV batteries, the inclusion of the pack casing and other components can help to reach the target. For example, if the casing is aluminium and constitutes 30% of the mass of the battery pack, and can easily be separated from the cells, it gives a huge boost towards achieving the target. New pack designs have a higher cell-to-pack ratios (up to 85% by mass) meaning less of the mass is occupied by the casing and more by the cells, making it more difficult to achieve the targets.
Solvent recovery becomes important as it helps towards achieving the target but being able to use the recovered solvent in another application can be challenging as different batteries use different solvents and further distillation will be required. If the solvents contain fluoride ions (highly likely), then these must be removed before they can be used again. Wet processes make it more difficult to recover the solvents as they must be separated from the water as well as from each other and some of the solvents can react with chemicals in the water (such as LiOH generated from the Li reacting with water). It may not be economical to recover the solvents from a wet process. Processes that involve thermal treatment cannot recover the solvents and therefore they will struggle to meet the targets, especially for portable batteries. Thermal processes also destroy plastics and combust some of which can help towards the efficiency targets although recycling separator plastics is not easy given that plastics recycling requires a melting and extrusion process which is only suitable for plastics with far more rigidity.
The big low hanging fruit is the recovery of graphite. This can give a huge boost to the recycling efficiency and potentially increase the value of the black mass but efficient separation often requires thermal treatment to remove the solvents as they interfere with the chemistry of the separation process. The thermal treatment must be at medium temperatures (below 500C) to stop the graphite reacting with the oxygen in the metal oxides. Normally, it is not the shredder that ends up with the graphite but the black mass processor/refiner and it is they who decide what to do with the graphite. In the future, it is likely that graphite recycling will become common place as battery grade graphite has a high value ($4-6/kg).
Waste Generation
All processes generate waste. Waste is material that cannot be sold. Some wastes can be landfilled whilst other wastes can be taken away by another company either to recycle or dispose of at a gate fee (producer pays company a fee to treat/dispose of waste). The most common wastes from battery recycling processes are plastic wastes and electrolyte solvent waste. There is also waste generated from gas scrubbing and dust extraction operations. Most of the dust can be added back into the black mass. The gases generated from the shredding and drying processes require scrubbing and capturing. Scrubbing can produce products such as calcium fluoride which require disposal but as it is a common by-product of other industrial processes, plenty of routes exist for its disposal.
Most processes cannot recover a sellable solvent product as it contains too many impurities and is often laced with fluorides. This material is difficult to dispose of as the normal route of burning it in a waste to energy plant is not available due to the presence of fluorides. It may require specialist incineration which is a very costly undertaking.
Wet treatment processes generate contaminated water. This water can be treated in an in-house water treatment plant to produce a concentrated solution containing various chemicals such as electrolyte solvents, lithium compounds, fluorides and phosphates. This water can be treated with other chemicals to destroy, remove and reduce the number of contaminants. The remaining water may require tanking off site at a cost. Processes that involve a thermal treatment step do not generate much solid waste other than the by-products of the gas treatment process. The gas treatment must be very comprehensive as the variety of gases, vapours and compounds formed from the thermal treatment step will be far more complex than those from a dry or wet process.
Conclusions
The choice as to which process to choose is driven mainly by the following factors; ability to meet the current and future safety requirements, product quality, ability to meet efficiency standards and throughput. The safety requirements regarding exposure to black mass dust will outweigh anything else and wet processes are best placed to meet the requirements. Wet processes produce lower quality products that affects process economics, however, this may not play such an important role as the highest prices are paid by Asian buyers who want dry black mass as it is common to thermally treat the black mass in Asia. In Europe, the black mass is normally washed by the hydromet processor. It will soon be very challenging to export black mass outside of Europe and it will not be a viable option. Therefore, there may not be that much of a difference between the value of dry black mass vs wet black mass.
References
Hydrometallurgical Recycling of Black Mass - When Does it Become Profitable?
Hydrometallurgy is an essential part of battery recycling. It is the stage that comes in between shredding/sorting and cathode active material (CAM) production. The term hydrometallurgy refers to the technique of transferring solid metals or metal containing compounds into an aqueous phase using corrosive liquids such as acids or sometimes alkalis. Once in an aqueous phase, the metal ions can be separated from unwanted impurities (as well as each other) using selective extraction techniques. Finally, the purified metal ions can either be recovered as salts or metals.
The core technology used in the hydrometallurgical recycling of black mass comes from the mining industry where decades of optimisation has produced highly productive mechanical and chemical processes that are now being transferred to the recycling industry. The recycling industry has a number of potential advantages over the the mining industry where the feed comes in the form of various ores which contain small concentrations of the target metals, requiring enormous amounts of energy to dig out of the ground, ship to the refining plant (often in another part of the world), pulverise into small particles which require concentrating up before processing. In the recycling industry, the black mass is already in a powder form and the concentrations of valuable metals such as nickel, cobalt are far higher than those found in ores where the concentration of the target metal in the ore is often less than 1%. The hydrometallurgical recycling process for black mass can be set up to produce metals, metal salts or a mixed hydroxide precipitate (MHP). Figures 1 and 2 show typical process pathways for MHP production [Fig 1] and metal/salts [Fig 2]. Note that other pathways exist that could potentially provide more efficient pathways - these will be covered in a future post.
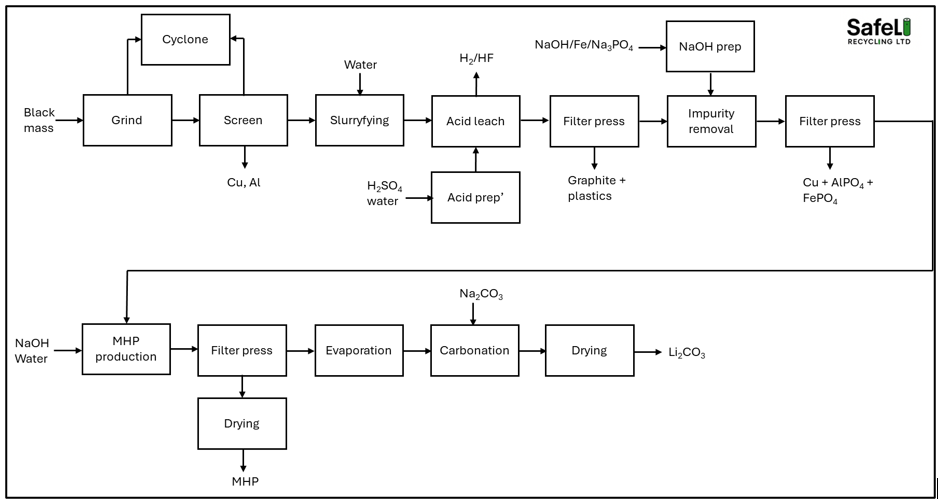
Figure 1. Simplified MHP process block diagram
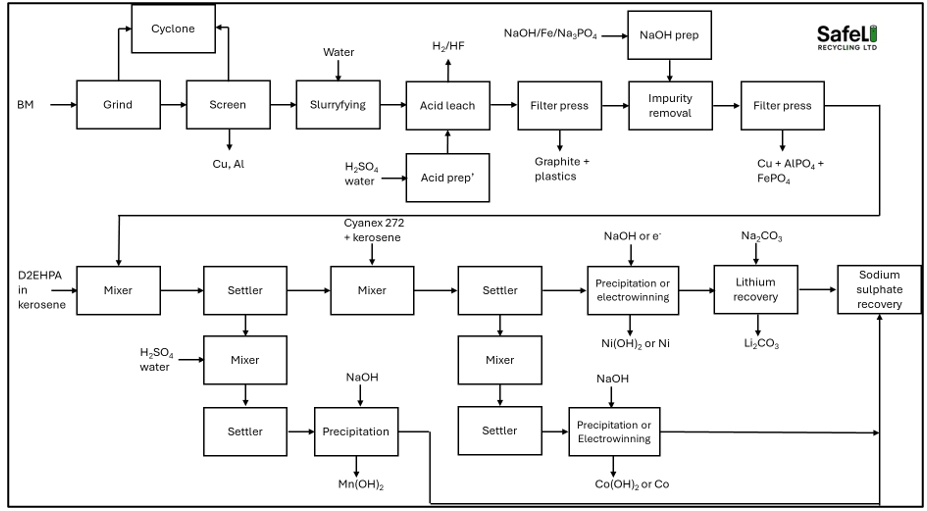
Figure 2. Simplified metal/salt process block diagram
Process Description
The black mass is first milled to a consistent particle size and sieved to remove metallic impurities such as copper and aluminium. Copper and aluminium impurities must be removed as they can contaminate the final product, affecting its value and potentially battery performance if the metals are recycled back into a battery. The black mass is then mixed with water to create a slurry that is pumped into a leach tank. Acid/water is added to the black mass slurry in sufficient quantities to make a ~2M solution. The addition of acid generates a number of gases as it reacts with the liquid, aqueous and metallic constituents of the black mass. The most hazardous gases formed are hydrogen fluoride and hydrogen gas. It is vital that LiPF6 is removed from the black mass prior to acid leach. This can be done through a thermal treatment process which thermally decomposes the LiPF6 or a washing and filtration step that removes the water-soluble constituents such as the hydrolysis products of LiPF6. A mere 1% LiPF6 concentration in the black mass can generate up to 7.9kg of HF per tonne of black mass. When sulphuric acid is present alongside the HF in an acidic medium, it generates an exothermic reaction, heating the mixture. The sulphuric acid acts to suppress the dissociation of HF and forms a very corrosive mixture that is far more corrosive than sulphuric acid alone. The HF within this mixture exists as HF rather than H+ and F-. This makes it extremely dangerous as it can enter the gas phase if heated, generating highly toxic fumes.
The hydrogen gas that is generated as result of the acid-metal reactions can form an explosive atmosphere and must be vented safely. A 1% metallic aluminium content can generate 13.6m3 of hydrogen gas (at STP) per tonne of black mass. The combination of copper and aluminium impurities together can generate a large volume of hydrogen gas which needs to be vented safely.
After the initial addition of H2SO4 and the subsequent calming of the reaction, a reductant such as hydrogen peroxide (H2O2) is added to aid the leaching process. Periodic measurements of the leach liquor are taken until most of the target metals are in solution (>95% leach). After this point, the pregnant leach liquor (the acid containing the metal species) is put through a filter press to separate out the solid undissolved material from the aqueous phase.
The next step in the process is the removal of impurities such as aluminium, copper and iron. To achieve this, the pH of the solution must be raised so that it is suitable for the process being carried out. The copper is either removed through a solvent extraction process or through the addition of iron powder. The iron powder displaces the copper at a pH range of 0.5-2. The aluminium and iron can be removed through the addition of sodium phosphate. Sodium phosphate is superior to using sodium hydroxide as it reduces the loses of nickel and cobalt.
The solid impurities are filtered out using a filter press and the aqueous phase is pumped to the next stage. For a plant producing MHP, the next step is the increase in pH to precipitate out the nickel, cobalt and manganese. These metals begin to precipitate out at a pH of around 6.5 – 10 with nickel precipitating out at the lower end and the manganese precipitating out at the higher end. After the solid MHP is separated out from the aqueous phase, the lithium can be precipitated out and recovered through the addition of lithium carbonate.
If the plant is designed to produce individual products (such as nickel hydroxide or nickel sulphate or nickel metal), it will need to use solvent extraction circuits. The solvent extraction stage is the most important part of the hydrometallurgical recycling process. The efficiency of the solvent extraction process is what makes or breaks a hydromet plant. The shorter the solvent extraction process, the more economical the process is and the more efficient the plant will operate with less downtime. The raffinate (aqueous phase from which a target metal has been removed) from stage one moves into stage two. In order for stage two to work optimally, stage one has to work optimally and in order for stage three to work optimally, stage two has to be optimal. If stage one does not operate optimally, stage two will not work optimally as any manganese remaining in solution will affect the extraction of cobalt. The greater the number of extraction stages, the more difficult it will be optimise the plant and the more difficult it will be to scale it up.
The manganese can be removed from the aqueous solution using D2EHPA (di-2-ethylhexyl phosphoric acid) diluted in kerosene. The extraction works best at a pH of 3.5-4. At this pH range, the D2EHPA forms strong bonds with manganese ions whilst nickel and cobalt remain in solution. The first stage is a mixing tank where the two phases (aqueous and organic) are mixed to allow the solvent to extract the manganese ions. Next the manganese ions in the solvent are transferred to a settler tank where the two phases separate out. The organic phase is then decanted and sent to another mixer tank where it mixes with a fresh batch of acid. The manganese ions transfer from the organic phase to the acid and the organic phase is recycled back to the mixing tank. The manganese in the acid can then be precipitated out as a manganese salt – either as a hydroxide or a sulphate.
The above process is repeated for the cobalt to separate it from the nickel. Cobalt and nickel have far higher values than manganese and refiners may choose to recover them as metals through electrowinning. The advantages of such an approach are the regeneration of the acid and production of high quality metal products that can be sold at full market value. The nickel and cobalt can also be recovered as sulphates through a crystallisation process but the sulphates will eventually require conversion into hydroxides if they are to be recycled back into a battery. After the lithium removal, the remaining sodium sulphate will require disposal. The most effective way to do is to crystalise it out as a solid and then pay for it removal or try and sell it as a product.
Economics of Hydrometallurgy
Setting up a hydrometallurgical plant is very expensive and operating it is also very expensive as it uses copious amounts of chemicals and energy and produces large amounts of waste. Currently, few black mass specific hydrometallurgical plants are turning over a profit from their operations. The rest of this article will try to show why that is.
To get sufficient economies of scale, hydrometallurgical recycling needs to be carried out on a large scale (>10,000 t/y). Most of the processes currently installed in Europe are either small scale operations or are demonstration plants. Some of the hydromet plants that have been installed in Europe are listed below;
- Primobuis, Germany – in partnership with Mercedes Benz. Capacity 6000t/y
- Fortrum, Finland – capacity 3000t/y
- Eramet, France– currently being built
- Euro Dieuze Industries (EDI), France – capacity unknown
- Umicore, Belgium – pyroàhydromet process. Capacity 7000/y LIB input
- Altilium Metals, UK – small scale facilities with CAM production capability
- Hydrovolt – no longer in operation
- AE Elemental, Poland – capacity unknown
- SungEel Hitech, Hungary – capacity unknown
Most of the operational hydromet plants produce technical grade products (<99.5% purity) and not battery grade products (>99.5%) as there is a global commodity market for the technical grade material. Battery grade products are technically very challenging to produce and the price difference may not be large enough to justify the additional expenditure and complexity in operations. In East Asia, where CAM manufacturing is more common, battery grade products are produced by some refiners, particularly those that are vertically integrated and have CAM manufacturing capability which allows them to absorb more of the cost of producing battery grade products as they can put extra resources into the purification process since their aim is to use the pCAM for CAM production. Therefore, as long as the cost of pCAM production is lower than or equal to the cost of the metals at market value, it is worth it. For companies producing only pCAM or technical grade metal salts, the cost of the producing the pCAM (battery grade or otherwise) must be sufficiently low so as to turn over a profit. This is the main weakness of hydrometallurgy because the margins are squeezed.
Lets start with 1000kg of NMC 6:2:2 black mass. The composition of the black mass is shown in Table 1. The total value in the black mass is around $7,415. If that black mass is purchased by the hydromet recycler at 50% of the value for Ni + Co totalling $2,720/t (this value is on the lower side, Asian buyers will pay far more), it leaves a margin of $4,595/t. If the refiner is only paying for the nickel and the cobalt, then lithium becomes the most important metal as the refiner is essentially getting it for free. Therefore, it stands to reason that lithium is the most important metal for the economics of the hydrometallurgical plant providing that the refiner can recover it efficiently and at an acceptable purity.
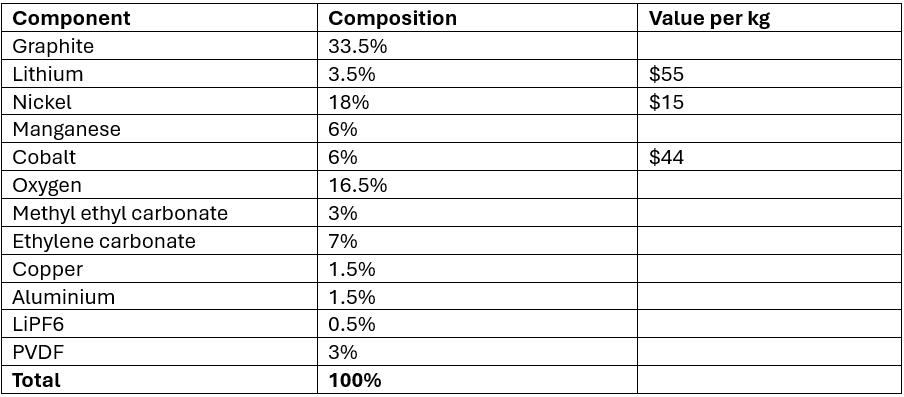
Table 1: Black mass composition
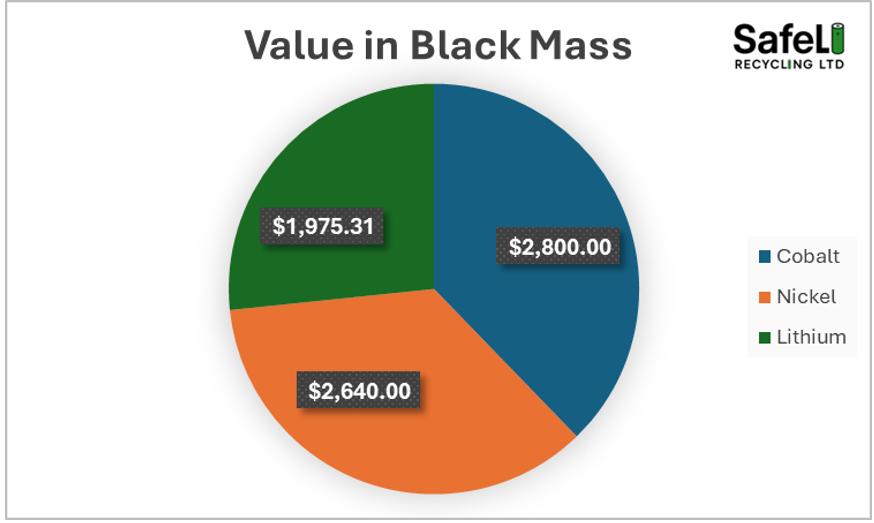
Figure 2. NMC6:2:2 black mass value
At what scale can a hydromet plant turn over a profit given the current prices for metals?
To answer this question, lets model a hydromet plant (in excel) producing MHP and lithium carbonate using the following assumptions;
- The base case for the plant is modelled on a 750t/y plant in the UK
- Increases in scale are achieved using commonly used scaling factors for labour, energy, space etc. These scaling factors can be found in literature and normally range from 0.4-0.9 with most falling between 0.6-0.7.
- MHP can be sold at 90% of the market value for Ni + Co (in reality, unless a battery manufacturer is the customer, the value on the open market is likely to be lower as the manganese dilutes the Ni and Co composition)
- The lithium recovery is 80% (as a carbonate) and is sold at 75% of its value
- Electricity is priced at $0.25/kWh
- Heating is done wit natural gas at a price of $0.067/kWh
- Waste treatment and disposal costs at $250 per tonne
- Assume plant availability of 85%
Cost of Processing as a Function of Throughput
Figure 4 shows the cost of processing the black mass using hydrometallurgy in the UK. The chart does not include financing costs and depreciation. As can be seen, at current prices for metals, the plant cannot make a profit even at 30,000t/y as the costs of processing are too high. At 30,000t/y, the plant will be making a loss of $570 on every tonne of material processed. The biggest costs are chemicals, followed by the gas and electricity use. A hydromet plant uses an enormous amounts of energy, for example, even for a simple MHP production plant, more than 3 tonnes of steam is required for every tonne of black mass. Raising that steam is very energy intensive and if the plant has to use electricity instead of natural gas, the costs of heating will be around 5x higher.
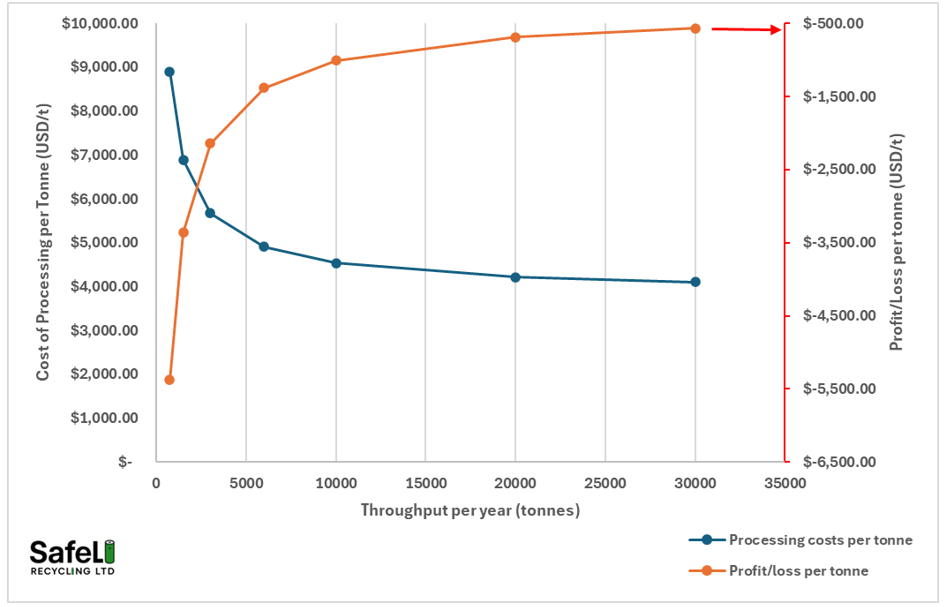
Figure 4. Cost of processing a tonne of black mass and profit/loss at given throughput using current prices for cathodic metals (Oct-2025)
Now lets assume a 20,000 tonnes per year plant. At what prices of cathodic metals does the plant begin to make a profit (not including financing and depreciation).
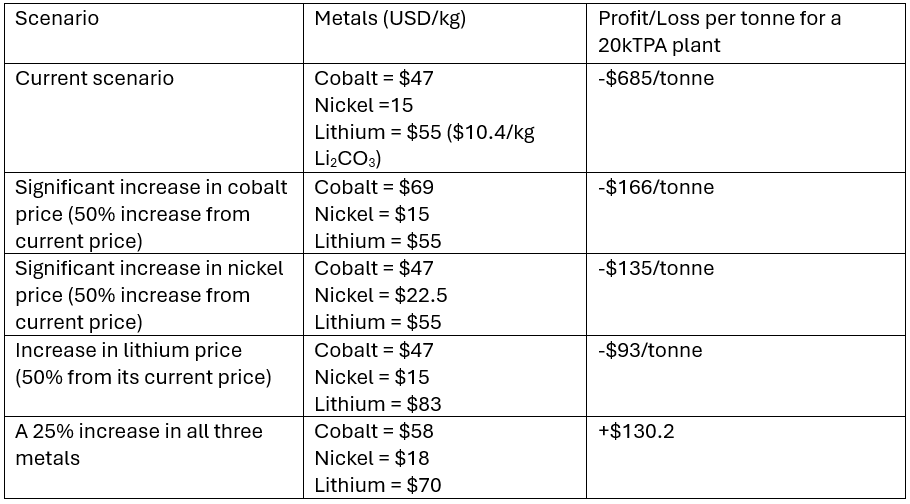
Table 2. Showing effects of metal price increase on profitability of the hydromet plant
From the results in Table 2, we can see that the price of all cathodic metals needs to increase by 25% before a 20,000t/y hydrometallurgical plant begins to make economic sense in the UK (if it is processing only NMC 6:2:2). The plant will likely operate tactically to make the most of the swings in metal prices. For example, in scenario 2, when the cobalt price increases, the plant can switch to a higher cobalt input such as more LCO in the feed to make the most of the higher cobalt prices.
In other parts of the world, the costs of operating such a plant will be significantly lower since labour and energy costs are far lower and overheads and admin costs will be less. The price of chemicals could also be less since countries like China have huge ecosystems for chemical manufacturing and all sorts of chemicals are available locally. In the UK, some chemicals have to imported into the country as they are not produced locally, increasing costs.
In terms of energy costs, in China for example, the electricity cost is around $0.11/kWh for industrial users or less than half the cost of industrial users in the UK. Furthermore, environmental legislations mean that UK (and European) industries will not be able to make use of natural gas for heating purposes. Therefore, if the plant requires 1000kg of steam, it will need to use around 915kWh of energy to raise it. If the steam is generated using gas, it would cost a UK company $61 but if it is generated using electricity, the cost rockets up to $228. A Chinese company raising the same amount of steam will be paying around $0.049/kWh, costing only around $45.
Finally, there is the cost of the plant itself. Hydrometallurgical plants are very expensive to set up. The most recent example is that of Li-Cycle. Li-Cycle spent over $900 million (USD) building its Rochester hub which was designed to process 35,000 tonnes of black mass per year. It is unclear why there were such enormous cost overruns but a 35,000t/y hydromet facility is still likely to cost between $500-600 million USD. Financing such a project can only be undertaken by companies with very deep pockets and should only be attempted once the pitfalls have been identified and ironed out through the building of smaller, pilot scale facilities. These facilities will most likely not be profitable but they will de-risk the project and save money and delays further on down the line.
Why Are Li-ion Battery Fires so Intense and How Do They Spread?
Lithium ion batteries offer the highest energy densities of any type of batteries and as such, they are used in an increasing number of applications. The down side with this is that it leads to an increasing number of battery fires since more batteries leads to a higher chance of fire. Since 2022, the number of call outs by the fire department for fires involving lithium ion batteries has increased from 600 to 1330 per year in 2024 (source: Money Super Market). The recycling industry is particularly vulnerable to battery fires as it is estimated that 48% of all waste fires occurring in the UK involve a lithium ion battery (National Fire Chiefs Council). So why is it that lithium ion batteries catch fire so readily and how does the fire spread?
By understanding the mechanisms that create battery fires and more importantly, how they spread, we can develop systems to counter them on recycling plants or other places where batteries are being stored.
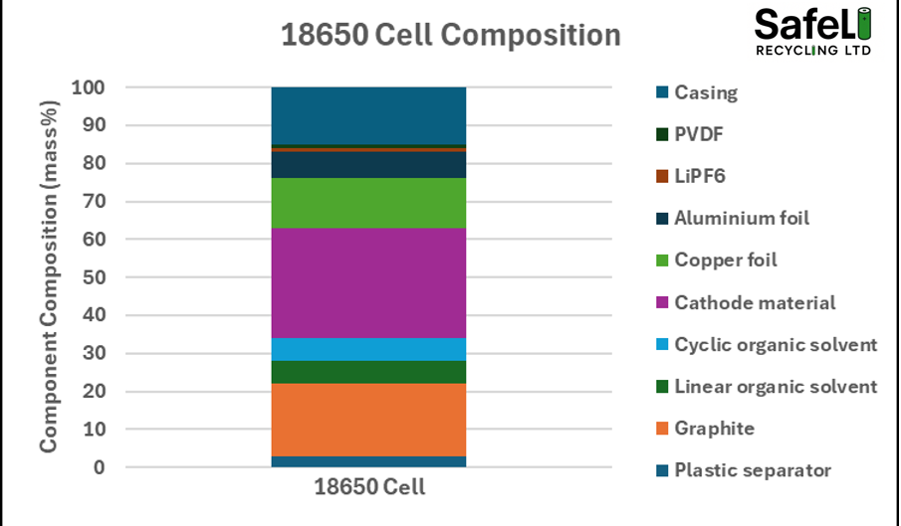
Before we get to the why they occur and how they spread, it is first important to consider the make up of a battery. Figure 1 shows that a typical 18650 lithium ion cell contains a variety of different components. Crucially, over 40% of its mass is combustible (plastic, organic solvents, graphite, aluminium and lithium). The 40% figure is on the lower end for most cell formats as other, more efficient formats such as pouch, can reach up to 50% combustibles within the cell. Notice also that over 10% of the mass of the cell is oxygen. In normal conditions, that oxygen is tied to the cathodic metals (such as nickel and cobalt), but it can become available for reaction if the conditions are right.
Figure 1. Typical 18650 cell composition
The next thing to look at is the energy that is present inside the cells. An18650 cell contains around 13Wh of electrical energy (3.5Ah @ 3.7V), however, this is not the only type of energy present within the cell. Remember that a battery is an electrochemical device as it converts chemical energy into electrical energy and vice versa. So, to enable that 13Wh of electrical energy, a lot chemicals energy must be present. Just how much chemical energy is present is shown in the table below.
Table 1. Chemical energy present in a cell
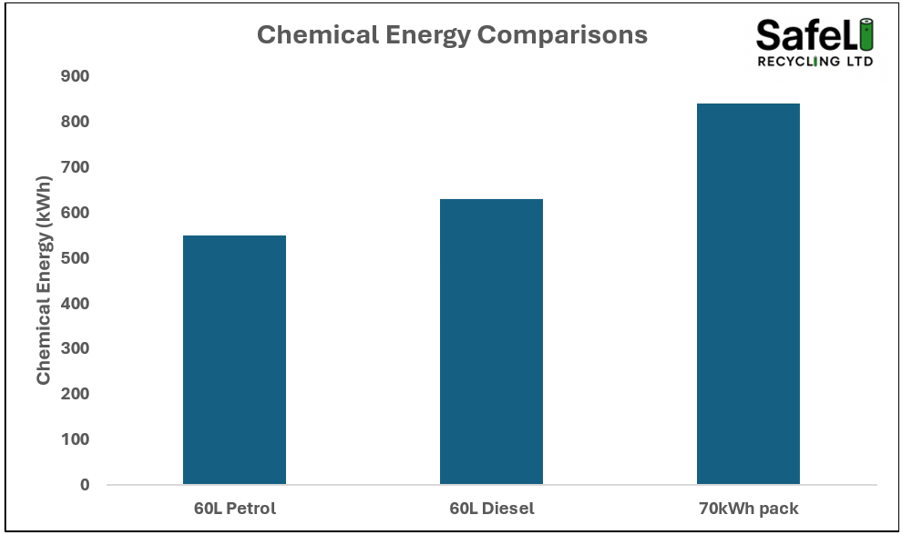
A typical 18650 cells weighs around 45g. 1kg of such cells would contain 22.22 cells with a total electrical energy content of 288.9Wh. To enable those 1kg cells to work, they contain the equivalent of 12.4 MJ or 3,444Wh of combustible chemical energy. The chemical energy dwarfs the electrical energy by a factor of 12.
To put this into perspective, a medium sized 70kWh battery pack will contain upwards of 840kWh of chemical energy. The chart below shows that a battery pack contains more combustible chemical energy than typical petrol and diesel tanks.
Figure 2. Chemical energy comparisons between petrol, diesel and a 70kWh pack
How Does a Battery Fire Start?
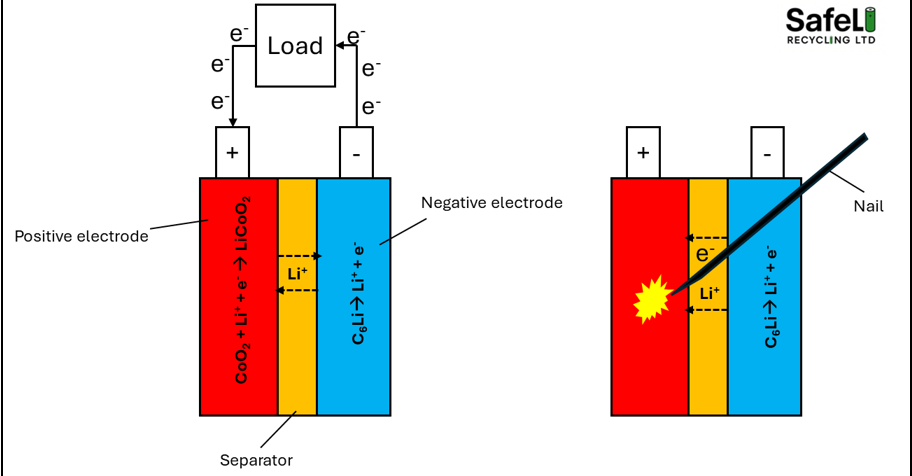
Now that we know how much combustible energy is inside a battery, the next task is to determine how a fire starts. There are various mechanisms as to how a battery fire can start, but once it gets going, the mechanisms by which it propagates, firstly inside the cell and then to other cells is very similar. Typical mechanisms by which a battery fire starts are as follows; short circuits, overcharging, storing in high temperatures, degradation, damage to the battery and in some cases failure of the safety systems – both internal and external.
A lithium ion battery works by lithium ions shuttling between the negative and positive electrodes (anode and cathode). When we charge a battery, we force the lithium to travel from the positive electrode, where they are stored in a stable structure (i.e. LiCoO2) to the negative electrode where they nestle within the graphite particles forming a highly unstable structure (C6Li). This structure gets more and more stressed with increasing state of charge and wants to lose the lithium as soon as it can. However, it cannot lose the lithium as it requires a pathway for the electrons to be able to travel from the negative to the positive electrode since the separator allows the lithium ions to flow through but not the electrons.
In normal operations, we provide this pathway by connecting it to an electrical device (a load bank). The electrical device takes the the energy out of the electrons, allowing it work. Once they go through the device, they come out at a much safer, lower energy state which can go back to the positive electrode without much heat build-up. If the electrons were to flow from the negative to the positive electrode without passing through a load bank, they would arrive at the positive terminal in a high energy state and would need to expend the energy. This is what we call a short circuit and it generates enormous amounts of heat energy, creating sparks.
Figure 3. Battery operation and short circuit mechanism
A short circuit is when there is an uncontrolled flow high energy state electrons (charge) from the negative electrode to the positive electrode. It is the most common cause of fires in lithium ion batteries and can occur in three ways;
- Internal short circuit
- External short circuit
- Damage to the battery
Internal short circuits normally occur due to a build up of dendrites (needle like metalloid structures) that form over time as the battery is cycled or if the battery contains metallic impurities that interfere with the electrochemical functioning of the cell. Eventually these dendrites can grow large enough to puncture through the separator, causing an internal short circuit. An external short circuit occurs when a conductive medium is placed across the two terminals. This leads to an uncontrolled flow of charge from the negative terminal to the positive terminal, generating heat and sparks.
Damage to the battery can lead to internal short circuits if the separator is broken, allowing charge to flow from the negative to the positive electrode.
In all cases, the result is a quick build up of heat that cannot be dissipated at a rate faster than it is being generated. Internal short circuits in particular are very dangerous as the heat is generated inside the cell and may not be noticeable until it is too late. This is especially poignant if the cell is buried deep within a barrel. The build up of heat begins to volatilise the electrolyte, increasing the pressure inside the cell. Further heat build up leads to the break down of the electrolyte components into smaller gaseous species such as CO2, CO, H2, etc., further increasing the pressure. The increase in temperature is associated with an increase in gas volume as the two share a proportional relationship, hence more pressure. As the internal short circuit continues, it generates more heat which begins to destabilise the cathode metal oxide species and above 200C, oxygen may be released by some of the cathode species. This oxygen becomes available to react with the electrolyte components, turbocharging the heating and reaching temperatures as high as 800C. The pressure inside the cell becomes too much to bear for the casing and it bursts open expelling a plume of superheated gases and vapours (mostly made up of electrolyte components) into the surroundings.
The expelling of the gases and vapours is what leads to the spread of the fire. In some cases, if the cells are small enough, the force of the gaseous ejection propels the burning cell in the opposite direction to the ejection and into a fresh batch cells where it continues to burn and heat up other cells, destabilising them and forcing them into a thermal runaway through similar mechanism to those described above. The initial gases and vapours released by the expulsion are already above their auto-ignition point and as soon as they mix with a sufficient quantity of oxygen, they burst into flames. Vapours released after the initial expulsion may not necessarily burn and in many cases, they exit the cells and travel along the ground (they are heavier than air so sink to the floor) creating an eerie fog-like effect, generating an explosive atmosphere which can ignite at any moment.
Fire fighters are reluctant to enter spaces where a lithium ion battery fire is still raging as many of them have experienced this fog-like substance. This reluctance can cause friction with the plant owners who want to save their plant/stock. Speaking with fire fighters who have responded to battery fires in the contained spaces, they mentioned that there were two distinct products from the fire; one, a black smoke that rises to the top (combustion products) and the other, a white fog-like substance that in some cases, they had to wade through to get to the fire. The toxic gases coming off of the fire adds further danger to the scenario.
The batteries will continue to burn so long as fresh cells are available to burn. Water is an adequate extinguishing agent as its high heat capacity and latent heat allows for enormous amounts of heat energy to be taken away from the fire, providing a cooling effect and stopping the destabilisation of newer cells and the spread of the fire. However, even if the fire is put out, the electrolyte components inside the cells have a low boiling point and continue to evaporate, generating copious amounts of vapour that can cause explosives atmospheres.

Lorem ipsum dolor sit amet, consectetur adipiscing elit. Nulla euismod condimentum felis vitae efficitur. Sed vel dictum quam, at blandit leo.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Nulla euismod condimentum felis vitae efficitur. Sed vel dictum quam, at blandit leo.
Lithium Recycling from Black Mass - Should it be First or Last?
Lithium is the most essential element that goes into a lithium ion battery (LIB). It has the perfect properties for an electrochemical storage material; it is the lightest metal in the periodic table enabling light weight batteries which are required for both automotive and portable applications (think lead acid battery Vs a lithium ion battery). It has the most negative electrode potential (vs a standard hydrogen electrode) of any metal, allowing it to maximise cell voltage. It forms a single charge ion (Li+), avoiding complex chemistries and side reactions associated with ions of more than a single charge. It also has many other benefits which I will not list here.
Making battery grade lithium is no easy feat. The majority of the lithium is mined in Australia and South America, but most of the refining is done in China which controls around 72% of the world’s refining capacity [1]. China is also the largest producer of lithium ion batteries and hence the largest consumer of lithium but it also exports thousands of tonnes of lithium ion batteries to all corners of the globe. This allows for the localised recycling and production of lithium providing that the locality has the technology to do so.
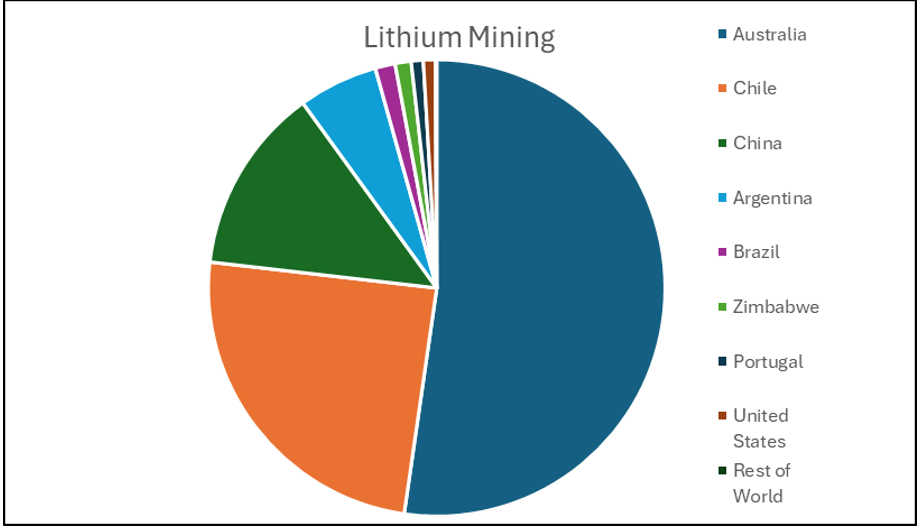
Chart showing where the world’s lithium came from in 2021 (data taken from World Economic Forum)
Battery Recycling
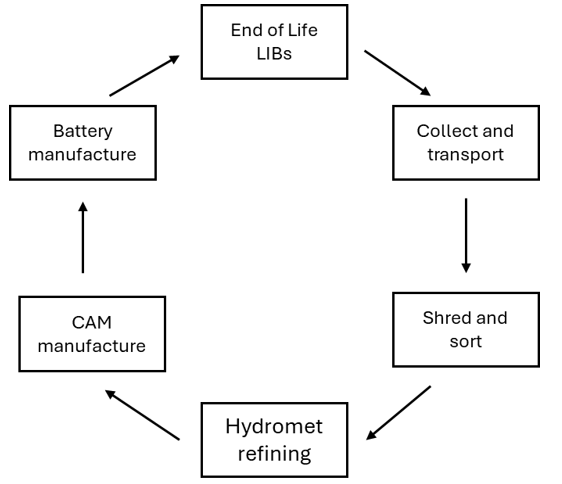
The battery recycling process is a five stage process. Stage one is the collection and transportation of the end of life batteries to a battery recycling facility. Stage two is the mechanical decommissioning of the batteries (shred and sort) to produce a number of products; copper, aluminium, steel and black mass – which is a mixture of the active powders that make the battery work (graphite and a lithium-transition-metal oxide such as lithium cobalt oxide). The third stage is the hydrometallurgical refining of the black mass to recover the valuable metals as well as the lithium in a salt form (such as nickel and cobalt hydroxides and lithium carbonate – these are known as precursor cathode active material or pCAM). Stage four is to use the recovered metal salts to produce cathode active material (CAM) and stag five is to use the CAM to make new cells that can be reintroduced into the market.
Being the most reactive and mobile metal within the battery, some of the lithium can be lost along the recycling process. For example, shredding plants that operate wet processes are likely to lose some of the lithium as it will react with the water. Some plants may use certain salts to improve the efficiency of the delamination process (taking the black mass off of the electrode foils) and these salts can interact with the lithium, making it more soluble and thus being lost in the water.
This focus of this article is on the hydrometallurgical recovery of the lithium. There are two competing processes – the first one is the standard route, which is that lithium is recovered last after all the other metals are recovered and the other is a lithium first route, where the lithium is the first metal to be recovered and the rest of the metals are recovered afterwards. As someone who has worked on and successfully developed (and patented) a lithium first process, I have my biases but I will try and keep these biases in check as I compare the two process routes. Lets start off with the standard lithium last route
Lithium Last Hydrometallurgical Route
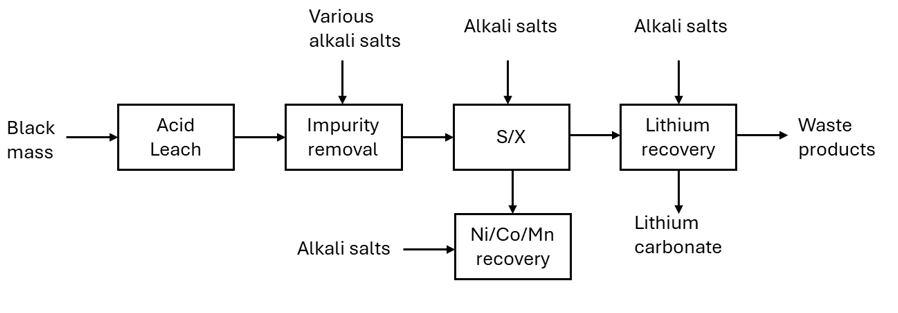
During hydrometallurgical processing, sulphuric acid is the standard acid used to dissolve black mass. More than 10 tonnes of (dilute) acid can be used for every tonne of black mass. Tonnes of various alkali metal salts (along with more water) are added to the pregnant leach solution (acid solution containing dissolved metal ions) to raise its pH, enabling the selective removal of different metals at different stages. Alkali metal salts are added to initially to increase the pH of the solution, they are then added to remove impurities such as aluminium and iron, more are added in the solvent exchange process to make the solvents have the right pH for extraction and finally, more alkali salts are added to recover the lithium. The added alkali metal salts all have similar properties to lithium salts and are all present in the solution when all the other cathodic metals (nickel, cobalt and manganese) are removed. When the lithium is precipitated out using sodium carbonate, some of these alkali metal salts also precipitate out with it and these act as impurities within the lithium, affecting its price. Standard hydrometallurgical processes can recover a technical grade (<99.5%) lithium carbonate that is not suitable for going back into the battery supply chain. The technical grade lithium carbonate must either be further upgraded, or it must be sold into the wider market where its price is heavily discounted against battery grade (>99.5%) lithium carbonate.
Lithium First Route
The lithium first route takes the opposite approach to the standard method as it recovers the lithium before any of the other metals are recovered. In normal conditions, the lithium in the CAM is in a non-reactive state as it is already attached to the other CAM components. Lithium, being very reactive, is easily activated through the use of heat or certain reducing reagents or a combination of the two. Depending upon the conditions and reagents used, the lithium can end up as an oxide or a carbonate. Both of these forms can be recovered with water as the oxide reacts with water to generate lithium hydroxide which is highly soluble and the carbonate is also soluble in water. Once dissolved into water, the lithium salts can be recovered through a simple filtration step.
The following companies are known to have developed lithium first processes
- Cylib Recycling
- Ascend Elements (announced they were building a plant capable of recovering 3000t/y of lithium carbonate, but it is not known if the plant was ever built) [3]
- ACE Green Recycling
- Smaller companies that are developing promising routes such as Battery Minerals
Advantages of Lithium Last Process
The major advantage of the lithium last process is that it does not require an additional plant that uses a different type of process (for example a thermochemical process) to recover the lithium. The same type of equipment that is used to recover the other metals are also used to recover the lithium. This makes it much easier to obtain the right type of permits, maintenance of the plant will be easier as will the training of staff.
Advantages of Lithium First Process
Lithium first processes have many potential advantages over the lithium last processes. Some of these are listed below:
- The first major advantage is that it can be deployed as a stand-alone process – without the need for a full-scale hydrometallurgical plant. This makes it very attractive for black mass producers who want to extract as much value from the black mass as possible.
- The next advantage is that it uses far less reagents to recover the lithium. In lithium last processes, tonnes of acid and alkalis are used before lithium recovery is possible. In lithium first processes, the reagent use is minimised
- Higher purity – as there are no alkali metal salts required for impurity removal or acid neutralisation, the purity of the lithium can be higher. In a previous employment, my team at Warwick Manufacturing Group were able to extract 99.8% pure lithium salts from mixed chemistry industrial black mass (supplied by RS Bruce, UK)
- The next advantage is that lithium first processes offer higher recovery efficiencies as lithium is lost in every stage of the hydrometallurgical process. The more steps there are until the lithium recovery, the more lithium is lost. In lithium first processes, the number of steps before the lithium is recovered are minimal so the availability of lithium for recovery is higher and in lab tests, up to 95% of the stoichiometric lithium in the black mass can be recovered. This figure will decrease somewhat as the processes are scaled up but it will still be far higher than the 80% offered by lithium last processes.
- Another advantage of the lithium first route is that it upgrades the black mass – depending on the mode used, the metals in the black mass can be reduced into base metals which are far easier to leach into acid and require far less reducing agents such as H2O2 to get them into solution.
- Finally, another big advantage for the lithium first process is that depending on the mode, it is possible to generate lithium hydroxide which is the lithium salt required for higher nickel content chemistries, such as all the latest NMC and NCA type chemistries. To achieve the same with the lithium last processes, you would require additional treatment in an electrochemical plant.
Disadvantages of the Lithium Last Route
The major disadvantages of the lithium last route are that the purity of the recovered lithium salt is low and therefore commands a much lower price than battery grade lithium salts – can be up to 33% lower. It cannot generate lithium hydroxide in one step and requires additional processing in another plant to generate it. To recover the lithium this way, you would need a full scale hydrometallurgical plant which costs hundreds of millions of dollars and are very expensive to operate. The lithium recovery efficiency for such plants is low because with each processing step, lithium is lost – for example, during the impurity removal step, the precipitated impurities are removed using a filter press (or a similar device) to form a filter cake. The cake will have a certain moisture content – say 40%, this moisture will contain all the metals that are present in the acid. Most of these can be washed out of the cake but some will still remain and the washing makes the lithium concentration more dilute and harder to recover.
Disadvantages of Lithium First Route
The main disadvantage of the lithium first route is that it relies heavily on the price of lithium salts. In many ways the lithium last route is also the same but as other metals are also recovered, the low price of lithium can be shielded by the (potentially) higher price of the other metals such as cobalt and nickel. Another disadvantage is that if lithium hydroxide is formed, it can react with aluminium impurities within the black mass which can act as an impurity within the lithium and requires further processing to remove it (which can be done in the same plant).
OK, so the big question now is, when does a lithium first plant become attractive? To answer this question, we need to understand that black mass producers are not paid for the lithium in the current market. In the future it may be the case but as things stand, black mass pricing is based solely on nickel and cobalt values. Therefore, it is in the interests of the black mass producers to recover the lithium themselves. So, the question becomes the following; at what price for lithium does lithium first recovery become compelling?
The answer to this question will be different for different but we can look at the proportion of the value that is attributed to lithium in a tonne of black mass and work from there. At the current price of cathodic metals (Ni = $15/kg, Co = $33/kg and Li = $53/kg), if we take an NMC 622 black mass with 58% CAM and 35% graphite (with the rest being impurities), the lithium accounts for only around 28% of the total value of the black mass or around £1,650 ($2,200).
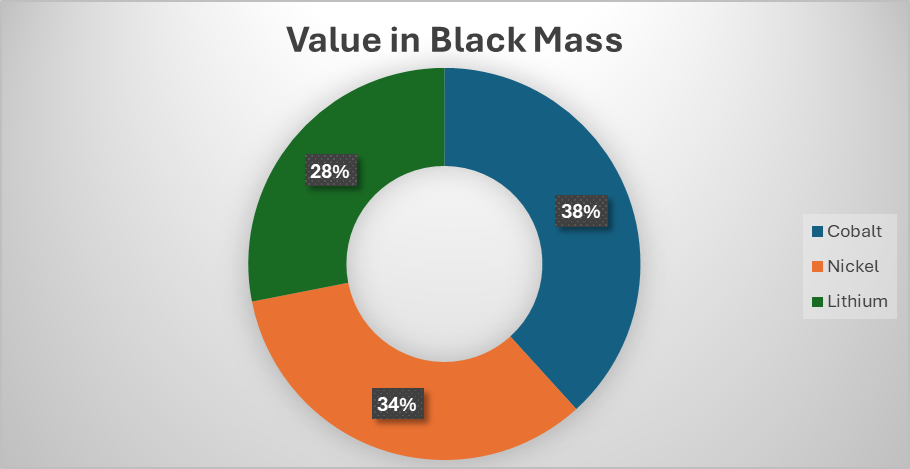
It is well known that over the last couple of years, there has been an oversupply of lithium on the market and the price of battery grade lithium products has consistently been below the price of production. This is the result of the lumpy investment model that operates in the mining sector. When there is a 10,000-tonne deficit of a certain commodity, the price shoots up, prompting new mines to open up. However, when a new mine opens, it doesn’t just add 10,000 tonnes of metal onto the market, it adds many times more than that, crashing the price until the demand catches up with the supply (or less profitable, high cost operations close).
In China, the production of battery grade lithium carbonate is thought to be around $18-20/kg. If we take this figure and add a 20% profit margin to it, we get to around $24 or $25/kg. According to Fastmarkets [3], in 2026, there is likely to be a slight lithium deficit and so lithium prices will reflect this but new supply is available so it won’t overshoot like it did in 2022 (lithium carbonate price has already risen by around 30% in the last couple of months). If we take $25/kg as the long-term benchmark for lithium carbonate ($132/kg for lithium) and also increase the prices of nickel to $20/kg and cobalt to $40/kg. The proportion of the value of lithium in the black mass increases to a whopping 49% of the total value of the black mass or £5,160 per tonne ($8,900). All this value will be lost to the black mass producer…unless they can recover it themselves via a lithium first process.
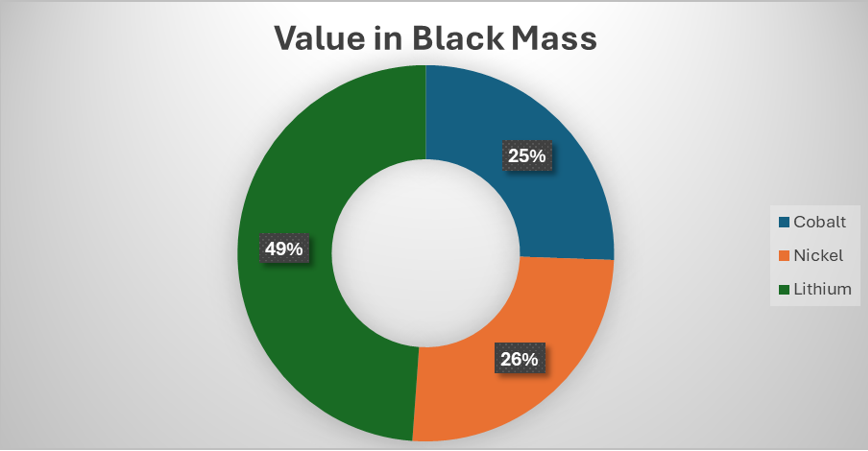
I have not yet worked out how much it will cost to make such a plant, however a medium sized operation processing 10,000 tonnes of black mass will cost between £25-30 million ($33-$40 million). Such a plant will produce around 1600 tonnes (assuming 85% plant availability) of lithium carbonate annually (assuming 90% lithium recovery efficiency), with a value of £30 million ($40 million). At those prices, lithium first becomes extremely attractive. In comparison, a hydromet plant processing 10,000 tonnes of black mass per year will cost well over $100 million with enormous operating costs.
If you found this article insightful and are interested in lithium first recovery processes or if you’d like to see how SafeLi Recycling Ltd can help you increase profitability or solve a chemistry/engineering problem, please get in touch.
